1. INTRODUCTION
Wetlands are ecotones between terrestrial and aquatic environments, and this transition gives them richness, functional diversity, and multiple values (Fustec & Lefeuvre, 2000; Maltby & Barker, 2009; Mitsch & Gosselink, 2007). Unfortunately, due to the anthropic effect applied to these ecosystems (Turner, 1992), they have become more vulnerable and/or degraded (Williams, 1990).
Among the anthropogenic activities, we cite the production of plastics, on which the world depends heavily. Because of its wide use in the industrial sector, which causes an increase in production (Rochman et al., 2013), it is projected that plastic production will reach between 902 Mt (Geyer et al., 2017) to 1124 Mt (Ellen MacArthur Foundation, 2017) by 2050. Within the various applications of plastic are construction, food and packaging industries, pharmaceuticals, and other sectors (Xu et al., 2019). Although plastic has many diverse uses, its drawbacks are even greater, leading to the pollution of ecosystems (Law et al., 2010; Ryan, 2013). Plastic can be divided into large particles and it can also break down into small particles that are only visible under a microscope; these are known as microplastics (Gall & Thompson, 2015). Microplastics are defined by their size, ranging from 5 mm to 1 µm (Hartmann et al., 2019), they have been found in marine environments, lakes, reservoirs, wetlands, estuaries, and even in polar regions (Auta et al., 2017; Eerkes-Medrano et al., 2015). Microplastics are categorized into primary and secondary types. Primary microplastics are intentionally manufactured and used in products like hand cleansers, facial cleansers, and toothpaste (Lassen et al., 2015). These are specifically produced by the plastics industry (Auta et al., 2017). In addition, primarily microplastics can enter wetlands through various routes, such as sewage discharge, surface runoff, and plastic waste (Qian et al., 2020; Reynolds & Ryan, 2017). Secondary microplastics are created when larger plastic fragments break down due to abiotic factors like intense solar UV radiation and mechanical abrasion (Gazal & Gheewala, 2020).
Algeria produced around 13.5 million tons of solid waste from households, with approximately 15.31 % being plastic, equaling about 2.07 million tons of plastic waste. Regrettably, only 15 % of this plastic waste is recovered (NAW, 2020, 2021). Production has risen in both Algeria and globally, especially during the COVID-19 pandemic. Every minute, about 3 million face masks are thrown away worldwide, contributing to environmental pollution (Prata et al., 2020).
The contamination of coastal and marine ecosystems has become a major environmental problem. Microplastics pose a danger to public health (Li et al., 2022). The Macta marshes were listed in the Ramsar Convention in 2001 as a wetland of international importance, as it is home to a wide variety of flora and fauna, and is a stopover site for migratory birds (Jacobs & Ochando, 1972; Lednat & Vandijk, 1977; Metzmacher, 1979). According to Véla & Benhouhou (2007), this area is a hotspot of biological diversity.
Microplastics can be checked in various environments, such as water (Luo et al., 2019), sediments (Uddin et al., 2021), soil (Harms et al., 2021), air (Gasperi et al., 2017), and living beings (Rillig et al., 2017; Zhang D. et al., 2020). Various scientific studies and research on microplastics have been conducted (Taïbi et al., 2021; Setiti et al., 2021; Amenouche et al., 2025; Grini et al., 2022; Devi et al., 2024; Rahmani et al., 2025; Khazr et al., 2025). However, no investigation has been conducted on microplastics in the Macta marshes, Algerian wetland. Our study is the first to examine the accumulation of microplastics in sediment and surface water at the Macta marshes, a Ramsar site in Algeria.
This paper aims to analyze the abundance and physical characteristics of microplastics, including their shapes, colors, and sizes, as well as their chemical properties (type), to help Algerian authorities and local environmental agencies build a successful program to reduce contamination emissions.
2. MATERIAL AND METHODS
2.1. Study area
The Macta Marshes have a triangular shape and are bounded to the north by the Mediterranean Sea. To the east, they are bordered by Mostaganem province. In contrast to the west, Marsat El Hadjadj Beach, and Oran province, and to the south, the mountain of Beni-Chougrane. Several rivers intersect this wetland, including the Sig, Habra, Tinn, and Oued El Hammam (Figure 1).
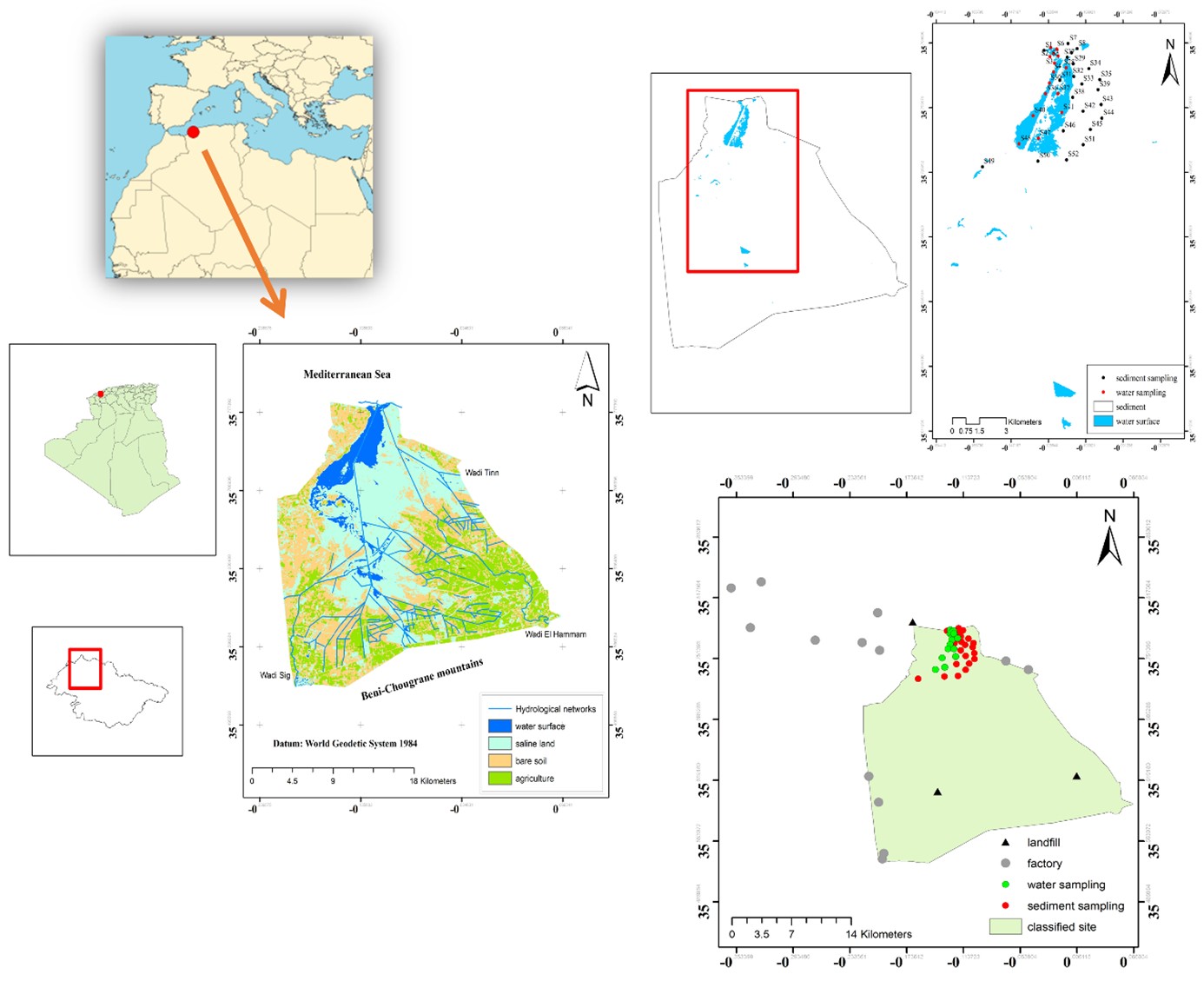
2.2. Sample collection
Samples for analyzing microplastics were obtained from sediments and surface water between March 07 to June 13, 2024. The specific latitude and longitude of the sampling site were determined using a Global Positioning System (GPS). A total of 22 stations were selected for sediment, and 15 surface water samples were collected from the Macta reservoir (Figure 1).
To collect sediment samples, a wooden quadrat measuring 25 cm × 25 cm (0.0625 m2) and a steel trowel were utilized to gather sediments from the top 3 cm of the surface. Sediment samples were taken in triplicate at each sampling station (Klein et al., 2015). Before sampling, both the quadrat and the steel trowel were rinsed with distilled water to avoid any risk of cross-contamination (Camargo et al., 2022). The gathered samples were wrapped in aluminum foil and stored at 4 °C until analysis (Ibrahim et al., 2021).
For surface water sampling, 1 liter of water was collected by steel containers from a depth of 0 to 10 cm at each site (Mercy et al., 2022). To enhance accuracy, three replicates were taken per site, resulting in a total of 45 liters of water per site. The samples were stored at 5 °C.
2.3. Laboratory process
2.3.1. Granulometric analysis
To determine the textural parameters of the Macta marshes sediments, two techniques of dry and wet sieving are used. The protocol described by Tnoumi et al. (2020) was employed.
2.3.2. Extraction of microplastics
The methodology established by Thompson et al. (2004) was implemented with modifications in the laboratory to isolate microplastics from sediments. Sediment samples were initially desiccated at 60 °C for 48 hours and subsequently sieved using a 5 mm stainless steel mesh to eliminate bigger, undesirable particles. Subsequently, 30 g of sediment was transferred into a 500 mL beaker, followed by the addition of 30 mL of hydrogen peroxide (H2O2, 30 %) to oxidize and eliminate natural organic waste. The mixture was left to react overnight. Afterward, 36 mL of zinc chloride (ZnCl2) solution (density ~1.58 g/cm³) was added for density separation. A magnetic stirring bar was added, and the mixture was stirred for 30 minutes. Then, the samples were left alone for 24 hours so that the different phases could separate. After that, the mixture was spun at 1500 rpm for 5 minutes to separate the solid from the liquid. Finally, a filter paper with a 0.45 µm pore size and a 47 mm diameter was used to filter the liquid (supernatant).
For microplastic extraction from surface water, we followed the method described by Wang et al. (2020) and Su et al. (2016), with some adjustments. Water samples were placed in 1000 mL Erlenmeyer flasks, and an initial filtration was carried out using a vacuum system and a 47 mm diameter filter with a 0.45 µm pore size for each sample. This step allowed for the retention of residues and microplastics in the water. After filtering, 100 mL of 30 % hydrogen peroxide (H2O2) was poured into a 250 mL wash bottle. The retained material on the filter paper was then gently rinsed into 250 mL glass flasks. The flasks were sealed with aluminum foil and heated in an oven at 60 °C for 48 hours to facilitate the reaction. Afterward, a second filtration was conducted using the same filter paper (47 mm diameter, 0.45 µm pore size).
2.4. Microplastics Identification
Microplastic identification was conducted according to the criteria established by Lusher et al. (2020). The filter paper was examined under a stereomicroscope at magnifications between 10x and 40x to analyze its shapes and colors (Pradit et al., 2022). We sorted microplastics into four groups: fragments, films, fibers, and pellets, using the method described by Hidalgo-Ruz et al. (2012).
There are 13 groups of colors: black, blue, gold, gray, beige, brown, green, pink, red, yellow, and white. Putting pink and raspberry in the pink group, orange in the red group, and a lot of other colors in the blue group, like bright blue, dark blue, magenta, purple, and cyan (Zobkov et al., 2020).
For size analysis, samples were examined using a scanning electron microscope (SEM). Microplastics were classified into two size groups: large microplastics (1 mm - 5 mm), small microplastics (1 µm - 1 mm) (Crawford & Quinn, 2016), and the polymer content of the samples was ascertained through FTIR analysis at a resolution of 8 cm-1, with measurements conducted across a spectral range of 4000 cm-1 to 400 cm-1.
2.5. Quality Control
The experiment was kept away from any outside plastic source to keep the microplastics from getting mixed up. They used cotton lab coats and gloves that didn't have any polymers in them (Jahan et al., 2019; Jiwarungrueangkul et al., 2021). The samples in the beaker were protected from any possible contaminants in the air by aluminum foil. All of the tools and glassware used during laboratory work were thoroughly cleaned and rinsed with distilled water. During the filtration process, personal filters were used to keep anything from getting in.
2.6. Statistical Analysis
Granulometric analysis (mean (Mz), Sorting (θ), skewness (Ski), and kurtosis (KG)) was calculated based on the methodology proposed by Folk & Ward (1957). The concentration of microplastics in sediments is measured as MPs/kg, while in surface water it is measured as MPs/L. This concentration is calculated as the average ± the standard deviation. A one-way analysis of variance (ANOVA) test was performed to assess the abundance, shape, color, and size of microplastics (MPs). When p-value less than 0.05 (p < 0.05) indicates a statistically significant difference, whereas a p-value greater than 0.05 (p > 0.05) suggests no significant difference. Statistical analyses were conducted using Excel 2016, and graphs were created using Origin 2018.
3. RESULTS
3.1. Textural characteristics
It is important to know the grain size distribution of sediments because they indicate the transport processes, level of weathering, and erosional features (Jian-Wu et al., 2013; Szcześniak et al., 2023; Tanabe et al., 2023; Boggs, 2006). The textural parameters for the Macta marshes are listed in Table 1. In which, the mean grain size (Mz) varies from 1.09 to 10.46 θ. The sediments are classified as: well sorted, moderately well sorted, poorly sorted, and very poorly sorted. The Macta marshes sediments are very coarse skewed, coarse skewed, nearly symmetrical, fine-skewed, and moderately fine-skewed. Kurtosis class range from mesokurtic to leptokurtic, very leptokurtic, and platykurtic.
|
Table 1. Textural characteristics for the Macta marshes sediments [mean (Mz), Sorting (θ), skewness (Ski), and kurtosis (KG)] (Folk & Ward, 1957). |
||||||||
|
Sample |
Mz (θ) |
Sorting (θ) |
Skewness (Ski) |
Kurtosis (KG) |
Mz (θ) Class |
Sorting (θ) Class |
Ski Class |
KG Class |
|
S1 |
1.26 |
0.52 |
-0.29 |
1.09 |
Medium sand |
Moderately well sorted |
Coarse skewed |
Mesokurtic |
|
S2 |
1.09 |
0.60 |
-0.18 |
0.96 |
Medium sand |
Moderately well sorted |
Coarse skewed |
Mesokurtic |
|
S3 |
1.69 |
0.55 |
-0.25 |
0.92 |
Medium sand |
Moderately well sorted |
Coarse skewed |
Mesokurtic |
|
S4 |
1.13 |
0.68 |
-0.69 |
1.10 |
Medium sand |
Moderately sorted |
Very coarse-skewed |
Mesokurtic |
|
S5 |
2.22 |
0.48 |
0.02 |
1.62 |
Fine sand |
Well sorted |
Nearly symmetrical |
Very leptokurtic |
|
S6 |
2.19 |
0.39 |
0.04 |
1.72 |
Fine sand |
Well sorted |
Nearly symmetrical |
Very leptokurtic |
|
S7 |
2.41 |
0.42 |
-0.03 |
1.73 |
Fine sand |
Well sorted |
Nearly symmetrical |
Very leptokurtic |
|
S8 |
1.53 |
0.86 |
-0.39 |
1.22 |
Medium sand |
Moderately sorted |
Very coarse-skewed |
Leptokurtic |
|
S9 |
1.66 |
0.59 |
-0.46 |
1.34 |
Medium sand |
Moderately well sorted |
Very coarse-skewed |
Leptokurtic |
|
S10 |
2.46 |
0.63 |
-0.16 |
1.20 |
Fine sand |
Moderately well sorted |
Coarse skewed |
Leptokurtic |
|
S11 |
2.03 |
0.69 |
-0.13 |
1.31 |
Fine sand |
Moderately well sorted |
Coarse skewed |
Leptokurtic |
|
S12 |
2.43 |
0.64 |
-0.4 |
0.99 |
Fine sand |
Moderately well sorted |
Very coarse-skewed |
Mesokurtic |
|
S13 |
2.10 |
0.49 |
-0.30 |
1.05 |
Fine sand |
Well sorted |
Coarse skewed |
Mesokurtic |
|
S14 |
1.37 |
0.92 |
-0.22 |
1.15 |
Medium sand |
Moderately sorted |
Coarse skewed |
Leptokurtic |
|
S15 |
8.78 |
1.29 |
0.35 |
0.82 |
Typical clay |
Poorly sorted |
Fine-skewed |
Platykurtic |
|
S16 |
9.69 |
1.48 |
0.46 |
0.75 |
Fine clay |
Poorly sorted |
Moderately fine-skewed |
Platykurtic |
|
S17 |
10.09 |
1.50 |
0.39 |
0.68 |
Fine clay |
Poorly sorted |
Moderately fine-skewed |
Platykurtic |
|
S18 |
9.34 |
1.83 |
0.51 |
0.87 |
Typical clay |
Poorly sorted |
Moderately fine-skewed |
Platykurtic |
|
S19 |
9.82 |
2.34 |
1.08 |
0.89 |
Fine clay |
Very poorly sorted |
Strongly fine-skewed |
Platykurtic |
|
S20 |
9.94 |
1.72 |
1.42 |
0.77 |
Fine clay |
Poorly sorted |
Strongly fine-skewed |
Platykurtic |
|
S21 |
10.29 |
2.10 |
0.45 |
0.73 |
Fine clay |
Very poorly sorted |
Moderately fine-skewed |
Platykurtic |
|
S22 |
10.46 |
2.62 |
1.21 |
0.81 |
Fine clay |
Very poorly sorted |
Strongly fine-skewed |
Platykurtic |
3.2. Analyses of microplastics in sediments
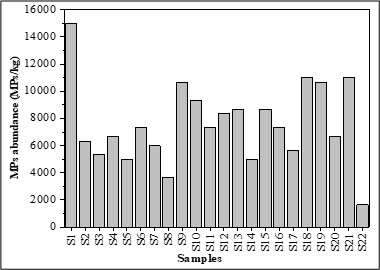
The sediment samples contained an abundance of 253.54 ± 273.84 MPs kg-1 of dry sediment (Figure 2). ANOVA showed no significant difference (p = 0.99 > 0.05, F = 0.29).
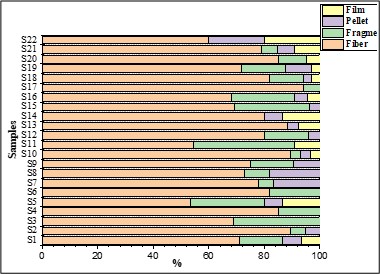
Regarding shapes (Figure 3), fibers were the most prevalent shape in sediments (80.73 %), followed by fragments (11.39 %), pellets (4.50 %), and films (3.37 %). The one-way ANOVA results for the shapes in the sediment samples indicated no statistically significant differences (p > 0.05), with F = 0.26.
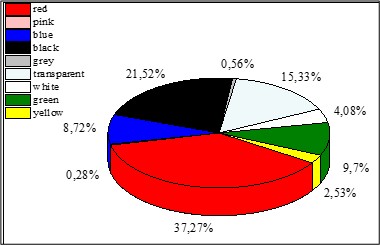
Our results on the color distribution indicated the presence of various colors (Figure 6). The most common color in the sediment samples was red, which accounted (37.27 %) of the total. The other colors in order, were black (21.52 %), transparent (15.33 %), green (9.70 %), blue (8.72 %), white (4.08 %), yellow (2.53 %), gray (0.56 %), and pink (0.28 %) (Figure 4). One-way ANOVA of color revealed no significant difference in the color of microplastics (MPs) (p > 0.05, F = 0.77).
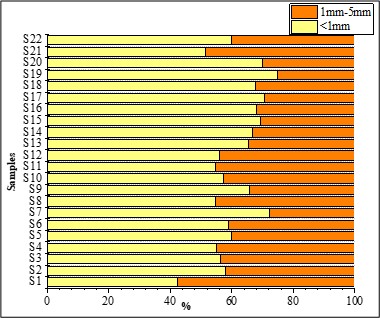
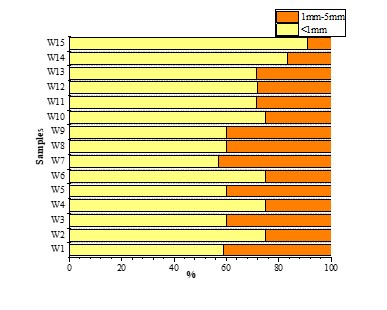
With SEM (Figure 7), we found that small microplastics were more prevalent than large microplastics.
Small microplastics accounted for 60.62 % of the total, whereas large microplastics accounted for 39.38 %, the results for each sample in Figure 5. One-way ANOVA for size showed that sediments (F = 0.23) were not significantly different (p > 0.05).


3.3. Analyses of microplastics in surface water
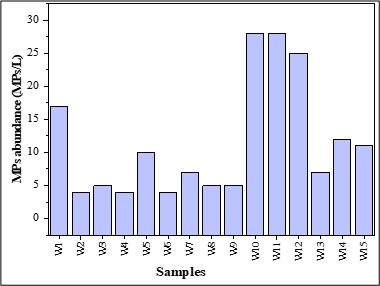
The abundance of MPs in the surface water samples was 3.82 ± 3.14 MPs L-1. The abundance of each sample of surface water in Figure 8. ANOVA showed no significant difference in abundance (p = 0.29 > 0.05, F = 1.24).
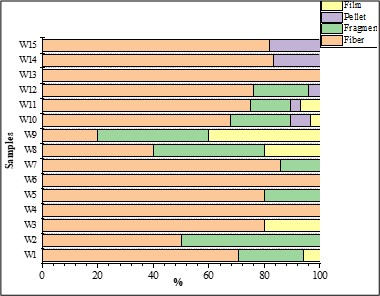
Fibers were the most prevalent shape (74.41 %), followed by fragments (16.27 %). Pellets (4.65 %) and films (4.65 %). Results obtained for each sample are presented in Figure 9. The one-way ANOVA results for the shapes in the surface water samples indicated no statistically significant differences (p > 0.05), with F = 0.83.
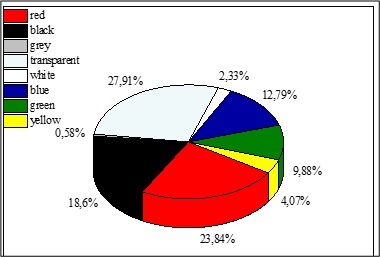
Transparent microplastics were the most common microplastics in surface water samples (27.91 %). The abundance of red microplastics is next (23.84 %), followed by black (18.60 %), blue (12.79 %), green (9.88 %), yellow (4.07 %), white (2.32 %), and grey (0.58 %) (Figure 10). There was a significant difference in the colors of the surface water (p < 0.05, F = 2.75).
Small microplastics (70.93 %) were found in surface water, but large microplastics (29.07 %). The results corresponding to each sample are illustrated in Figure 11. One-way ANOVA for size showed (p > 0.05, F = 0.38) that the size was not significantly different.
3.4. FTIR analysis
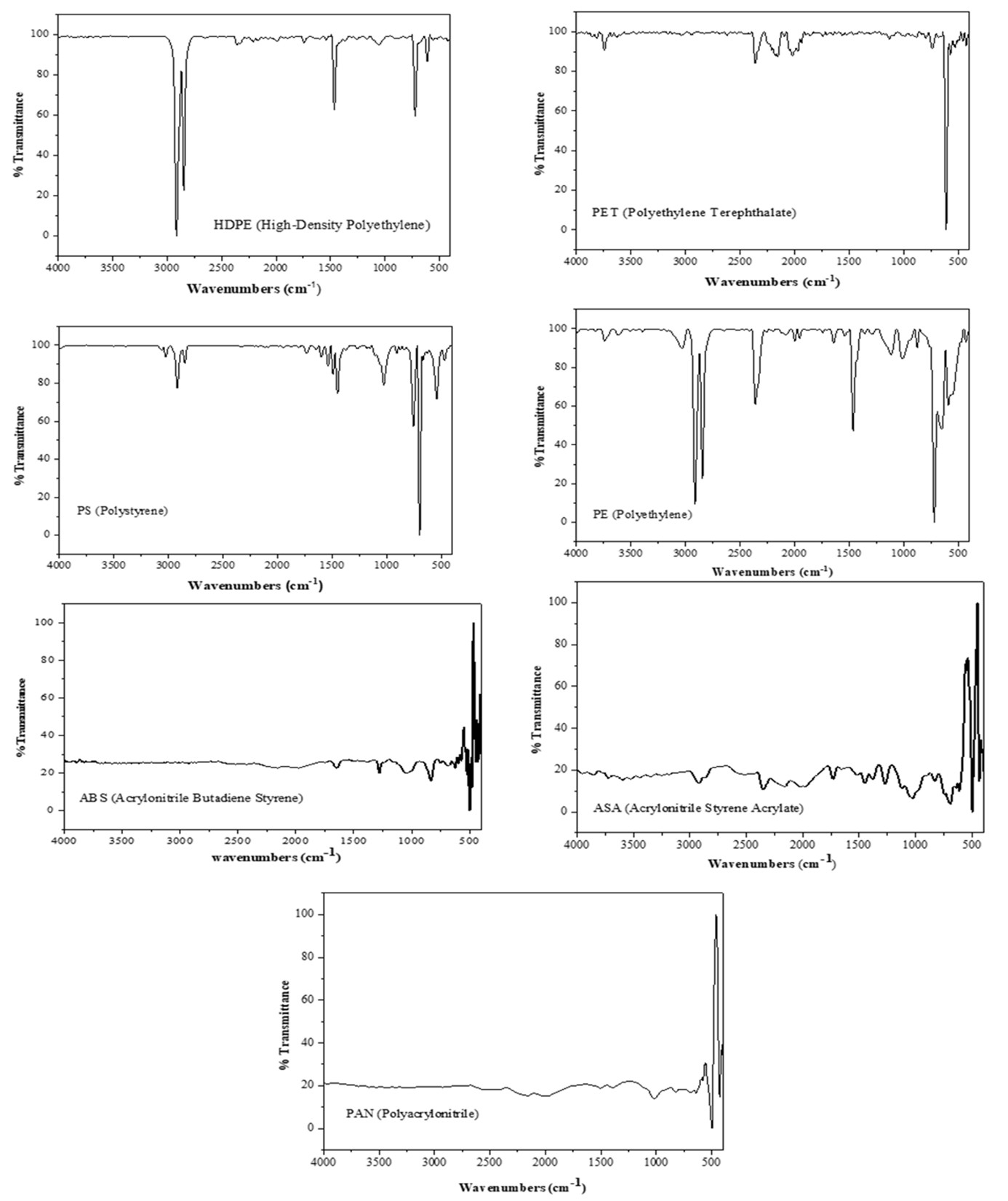
To determine the types of polymers present in the Macta marshes, we selected microplastics ranging from 1 mm to 5 mm to facilitate identification using FTIR techniques. Comparing of our data and spectra with the literature and spectra of other studies (Table 2), the results indicate the presence of several polymer types: HDPE (High-Density Polyethylene), PET (Polyethylene Terephthalate), PS (Polystyrene), PE (Polyethylene), ABS (Acrylonitrile-Butadiene- Styrene), ASA (Acrylonitrile-Styrene Acrylate), and PAN (Polyacrylonitrile) (Figure 12).
|
Table 2. Comparison of microplastic abundance in sediments and surface waters with global, Algerian, and nearby regional studies. |
|||||
|
Location |
Shape |
Size |
Sample type |
Polymer type |
Reference |
|
Gulf of Tehuantepec, Mexico |
Fibers, fragments |
< 5 mm |
Sediment |
PAM, PMA |
Ramos-Vázquez et al. (2024) |
|
|
|
|
|
|
|
|
Barra Norte and Tuxpan, Mexico |
Pellets, lines, fragments, films |
0.95 mm - 1.05 mm |
Sediment |
PC, PET |
Pérez-Alvarado & Armstrong-Altrin (2025) |
|
|
|
|
|
|
|
|
Pearl River Estuary, Hong Kong |
Fragments, pellets |
0.315 - 5 mm |
Sediment |
Expanded polystyrene (EPS) |
Fok & Cheung (2015) |
|
|
|
|
|
|
|
|
Five urban estuaries of KwaZulu-Natal, South Africa |
Fibers, fragments, films, pellets |
20 - 5000 μm |
Sediment & Water |
Not identified |
Naidoo et al. (2015) |
|
|
|
|
|
|
|
|
Al Hoceima Bay (Morocco), (near Algeria) |
Fibers, fragments, pellets, films |
0.15 - 5 mm |
Surface water |
PE, PP, PS, PET |
Bouadil et al. (2024)
|
|
|
|
|
|
|
|
|
El-Mellah Lagoon, Algeria & Bizerte Lagoon, Tunisia (near Algeria) |
Fragments (dominant), fibers (El-Mellah ~38 %) |
< 5 mm |
Sediment |
PE, PET, PP |
Khazr et al. (2025) |
|
|
|
|
|
|
|
|
Western Algerian Coast (Mostaganem-Arzew-others) |
Fragments, pellets, films |
≥ 1 mm |
Sediment
|
Not identified |
Taïbi et al. (2021) |
|
|
|
|
|
|
|
|
Bou-Ismail Bay, Algeria |
Fibers fragments films, foams, granules |
smaller sizes < 330 µm |
Surface water |
PE, PP, PS |
Setiti et al. (2021) |
|
|
|
|
|
|
|
|
Bou-Ismail Bay, Algeria |
Fragments dominant; Sediments: fibers dominant |
< 5 mm |
Surface water & sediments |
PE, PP, PS |
Amenouche et al. (2025) |
|
|
|
|
|
|
|
|
Skikda coast beach, Algeria |
Fragments, pellets |
1 - 5 mm |
Sediment |
Not identified |
Grini et al. (2022) |
|
|
|
|
|
|
|
|
Macta Marshes, Algeria |
Fibers, fragments, pellets, film |
(1 mm - 5 mm) (< 1mm) |
Sediment and Surface Water |
HDPE, PET, PS, PE, ABS, ASA, PAN |
Current Study |
|
Caption: polyethylene (PE), polypropylene (PP), polystyrene (PS), polyethylene terephthalate (PET), cellophane (CP), high density polyethylene (HDPE), polyvinyl chloride (PVC), Expanded polystyrene (EPS), polyacrylonitrile (PAN), polycarbonate (PC), polyacrylamide (PAM), polystyrene acrylonitrile (SAN), Low-Density Polyethylene (LDPE), Acrylonitrile Butadiene Styrene (ABS), Acrylonitrile-Styrene Acrylate (ASA). |
|||||
4. DISCUSSION
Several studies reported a significant correlation between MPs and the fine fraction of sediments: when the abundance of MPs increases, grain size decreases (Browne et al., 2011; Alomar et al., 2016; Mendes et al., 2021; Flores-Cortés & Armstrong-Altrin, 2022; Rodrigues et al., 2024). Conversely, both the research conducted by Ramos-Vázquez et al. (2024) and our own findings (P = 0.53, Pr = 0.47) show that there is no significant correlation between the presence of MPs and grain size. This is attributed to the fact that the distribution and accumulation of microplastics (MPs) are affected by particle density and environmental exposure (UV).
Our results indicate that microplastic concentrations are higher in sediments than in surface water. Woodall et al. (2015) suggest that this is due to the higher weight and density of microplastics compared to water, causing them to sink and accumulate in sediments. Moreover, the present study reports a higher abundance of microplastics (MPs) in sediments and in surface water than the results obtained in other studies (see Table 3).
|
Table 3. Studies conducted on microplastics worldwide, in Algeria, and in neighboring regions. |
|||||
|
Location |
Component |
Abundance |
Sampling methods |
Extraction methods |
Reference |
|
Mangrove Wetlands/China |
Sediment |
Inside: 429 items/kg; Outside: 21745 items/kg |
(top 2 cm) |
Density separation by CaCl2 |
Li J. et al. (2018) |
|
|
|||||
|
Anzali Wetland/Iran |
Sediment
Surface water |
June: 7836 items/kg; January: 5196 items/kg June: 1.77 items/m3; January: 1.25 items/m3 |
Van Veen grab (0.2 × 0.2m) Plankton net (350 μm mesh size) |
Density separation by NaCl |
Rasta et al. (2020) |
|
|
|||||
|
Three Wetlands: Portugal, Guinea, Bissau |
Sediment Surface water |
5970 items/kg 360 items/m3 |
Ponar grab (229 × 229 mm), Bulk (120L) |
Density separation by NaCl |
Ibrahim et al. (2021) |
|
|
|||||
|
Kallar Kahar Wetland/Pakistan |
Sediment Surface water |
5720 items/kg 88000 items/m3 |
Garden shovel Bulk (2.5 L) |
Digestion by H2O2 Density separation by NaCl |
Dilshad et al. (2022) |
|
|
|||||
|
Ashtamudi Lake/India |
Sediment Surface water |
3 ± 1.69/kg 2.75 ± 1.48/L |
steel trowels stainless-steel |
Digestion by H2O2 Density separation by NaCl |
Devi et al. (2024) |
|
|
|||||
|
Joumine stream, Northern Tunisia (near Algeria) |
Sediment
Water |
18.2 ± 8.27/50g
8.87±3.94/L |
Stainless-steel spatula Glass bottles (2 L) |
Density separation by NaCl |
Rahmani et al. (2025) |
|
|
|||||
|
Bizerte Lagoon, Tunisia (near Algeria) |
Sediment |
73.4 items kg⁻¹ |
Stainless-steel corer |
Digestion by H2O2 Density separation by NaCl |
Khazr et al. (2025) |
|
|
|||||
|
El Mellah Lagoon, Algeria |
Sediment
|
75.7 items kg⁻¹
|
Stainless-steel spatula
|
Digestion by H2O2 Density separation by NaCl |
Khazr et al. (2025) |
|
|
|||||
|
Bou Ismail Bay, Algeria |
Sediment Surface water |
0.36 ± 0.2 items/g 1.16 ± 0.8 items/m³ |
Sediment cores from multiple stations |
KOH digestion for water; KOH + Density separation for sediments |
Amenouche et al. (2025) |
|
|
|||||
|
Bou Ismail Bay, Algeria |
Surface water |
0.86 ± 0.35 |
Manta trawl (330 µm) |
Visual sorting |
Setiti et al. (2021) |
|
|
|||||
|
Western Algerian coast |
Sediment |
7.6 ± 18.8 to 66 ± 107.3 items/m² |
Manual sediment sampling + sieving ≥1 mm |
Manual extraction by sieving and visual sorting |
Taïbi et al. (2021) |
|
|
|||||
|
Skikda coast, Algeria Beach |
Sediment |
6 174–6 183 items/m² |
- |
Sieving and manual separation |
Grini et al. (2024) |
|
|
|||||
|
Current study (Macta marshes)/Algeria
|
Sediment
Surface water |
16733.33 MPs/kg
172000 MPs/m3 |
steel trowels
Steel container |
Digestion by H2O2 Density separation by Zncl2 |
Current Study |
The observed differences can be attributed to several factors, including the geographical location of the study area, the sampling and extraction methods used. In our study, sediment samples were collected using a steel trowel, and surface water samples were collected using a steel container. The extraction process involved using zinc chloride (ZnCl2) and hydrogen peroxide (H2O2). In contrast, other studies (Table 3) have employed various sampling techniques, including Van Veen grabs, Ponar grabs; these two techniques may affect surface sediment layers, causing resuspension, vertical mixing of sediment strata, and potential loss of low-density particles, which can lead to underestimation of their abundance (Hidalgo-Ruz et al., 2012; Claessens et al., 2011). For box corers, conserve sediment stratification and limit disturbance, permitting a more accurate estimate of the microplastics that have built up in the surface layers (Vianello et al., 2013).
In surface waters, employing plankton nets with different mesh sizes selectively captures specific size categories of microplastics. Larger mesh sizes often fail to capture smaller particles and fibers, which frequently predominate in aquatic environments (Lusher et al., 2014). As a result, differences in methodology significantly influence the observed distribution, abundance, and characteristics of microplastics in both sediments and surface waters. Other extraction methods have been used, including calcium chloride (CaCl2), sodium chloride (NaCl), hydrogen peroxide (H2O2), and zinc chloride (ZnCl2).
The variations in results can also be attributed to environmental pressures in areas near heavily populated rivers, as these environments are closely associated with intense human activity. The urban and peri-urban areas generate large quantities of plastic debris and synthetic fibers originating from domestic wastewater discharges, where the river mouths are considered significant routes for penetration of pollutants into ecosystems (Lots et al., 2017).
At these stations of sediments and surface water, you can often see birds drinking water and looking for food. These sampling stations are influenced by various sources of pollution, including landfill sources: domestic waste and garbage dump (Figure 1).
Factory sources: industrial zones such as the Macta desalination station, and the Algerian-Omani Fertilizer Company (AOA Fertilizers) (Figure 1). Additionally, in the north of the Macta marshes, Mediterranean Sea: Marsat El Hadjadj Beach makes the area susceptible to anthropogenic activities such as fishing and tourism. The Bethioua mineral port is situated near the river mouth of Oued El Hammam. Gardel et al. (2022) say that river mouths are places where sediments and tiny pieces of plastic tend to build up because the water currents slow down.
SEM images show that the surface of microplastics has cracks, indicating variations in the transport environment. Furthermore, the variety of microplastic shapes also points to more than one source of origin (Guo et al., 2018; Li L. et al., 2018). Fibers are observed to be the most common type of microplastics in a number of wetlands, including the Anzali Wetland in Iran (Rasta et al., 2020) and the Ramsar Wetland in Ashtamudi Lake, India (Devi et al., 2024). This is because of the shedding of staple fibers from fabrics (Yang et al., 2022). The high abundance of fibers in the Macta marshes is likely attributed to the laundry wastewater, sewage, domestic, and industrial sources. Fibers are commonly released from textiles during washing. Jiang et al. (2018) also said that fibers are common and easy to break, which is why they are prevalent in the samples. When studying other types of plastic debris, it was found that fragments were the second most common type in the Macta wetland. The primary sources of both fragments and film are likely the degradation of various plastic products, such as bags, packaging, and containers (Derraik, 2002; Nor & Obbard, 2014; Zhang et al., 2015). The plastic industry usually makes pellets from raw materials. In addition to being used in cosmetics and personal care products, such as facial cleansers, they are also utilized in the automotive industry (Mani et al., 2015; Karlsson et al., 2018).
Camargo et al. (2022) found that red was the most common color in the sediment of the Pantanal Wetlands in Brazil, which is similar to what we found. The occurrence of colored microplastics is predominantly attributed to the degradation of various plastic products frequently utilized in everyday life (Zhang et al., 2015; Wang et al., 2017).
Microplastics that are transparent are the most common type of microplastics found in surface water. According to Gündoğdu & Çevik (2017), the most common type of microplastic was clear microplastics. This is probably because clear plastic items that can only be used once are so common. Also, weak product color may fade over time due to exposure to the environment or treatment with hydrogen peroxide during lab analysis (Yin et al., 2020). On the other hand, red microplastics might be harder to break down and tend to accumulate in sediments, where they break down more slowly because they don't get much light (Corcoran et al., 2009)
The higher prevalence of small microplastics can be attributed to several factors, including fragmentation processes, hydrodynamic conditions, and rates of decomposition. Scientific research shows that bigger pieces of plastic break down over time through physical, chemical, and biological processes. This creates smaller microplastic particles (Andrady, 2011). Several studies agree with what we found, which shows that smaller microplastics are more common near the coast. Crawford & Quinn (2016) said that small microplastics are the most common type because they keep breaking up and spreading out in the environment.
A study done in estuarine and wetland areas showed that small microplastics are more common because they can move around more easily and stay in water systems for longer periods of time (Zhang K. et al., 2020). In addition, their higher numbers may be because they are easier for aquatic organisms to use, which eat them and spread them throughout the ecosystem (Setälä et al., 2014).
For the types of MPs: PE is the most commonly used material for packaging and single-use items worldwide (Plastics Europe, 2020). This kind of dispersion happens because they are heavy enough to float in water, even in wetlands (Cózar et al., 2014)
Microfibers of PET are the main source of water pollution, according to Boucher & Friot (2017). This kind is used in plastic bottles and synthetic fabrics (Browne et al., 2011), while polystyrene (PS) mostly comes from things that are only used once and packaging that is thrown away (Rochman et al., 2013).
ABS and ASA are two types of plastics often used in construction and automotive applications (Strong, 2006; Menges et al., 2001). So, PAN is mostly used in kitchen appliances, textiles, and household goods (Russell, 2007; Rosato & Rosato, 2001). Based on our results, there is plastic pollution in the Macta marshes due to urban runoff, domestic sewage, and industrial discharges.
5. SUMMARY
Microplastic distribution in Macta marshes showed clear contrasts between sediment and surface water samples. Sediments contained significantly higher microplastic abundances with 253.54 ± 273.84 MPs/kg. In contrast, surface waters exhibited lower and more variable microplastic concentrations (3.82 ± 3.14 MPs/L). This difference is due to the higher weight and density of MPs compared to water, which causes particles to accumulate and tend to sink and settle in sediment.
In both matrices, fibers were the dominant shape, accounting for 80.73 % of particles in sediments and 74.41 % in surface waters, followed by fragments, pellets, and films.
Distinct color patterns were observed between sediments and surface waters: sediments were mainly dominated by red, whereas a higher proportion of transparent particles characterized surface waters.
SEM observations revealed a predominance of small-sized microplastics in both environments, while FTIR analysis identified diverse polymer types, including HDPE, PET, PS, PE, ABS, ASA, and PAN. These differences reflect the combined influence of particle properties, hydrodynamic conditions, and local anthropogenic activities. Overall, domestic, industrial, marine, and recreational activities were identified as the primary sources of microplastic contamination in Macta marshes.
6. CONCLUSION
The current study is the first to focus on abundance assessment, physical characterization (shapes, colors, size), and type identification of microplastics in sediments and surface water in a Ramsar site, the Macta marshes. Our results indicate that the Macta mashes has a high concentration of microplastics due to the impact of domestic, industrial, and marine activities. The dominant shape of microplastics is fiber. In sediment, the color is predominantly red, while in surface water, it is transparent. This abundance is attributed to domestic discharge and industrial wastewater, as well as fishing activities, maritime operations, and the release of textiles during washing and laundry in communities. Most microplastics were sized less than 1 mm. The results of an FTIR spectroscopy analysis showed the presence of HDPE (High Density Polyethylene), PET (Polyethylene Terephthalate, PS (Polystyrene), PE (Polyethylene) ABS (Acrylonitrile-Butadiene-Styrene), ASA (Acrylonitrile-Styrene-Acrylate), PAN (Polyacrylonitrile) which indicate that the packaging, plastic bottles, and synthetic textiles were possible sources of microplastics, therefore the local authorities should investigate plastic pollution and develop a program aimed at managing and reducing solid waste in Macta marshes.
ACKNOLDGEMNTS
The authors would like to thank the staff of the University of Ahmed Zabana, Relizane (Algeria), for their assistance with sample analyses, as well as the Bioprocess, SEM, and Analytical Chemistry Laboratories at the University of Malaysia Sarawak for their support with sample analysis and microscopy.
REFERENCES
- Alomar, C., Estarellas, F. & Deudero, S., 2016. Microplastics in the Mediterranean Sea: deposition in coastal shallow sediments, spatial variation, and preferential grain size. Marine Environmental Research, 115, 1–10.
- Amenouche, S., Mokrane, Z., Belhouchet, N., Keraghel, M.A. & Hamdi, B. 2025. A multi-compartment assessment from surface waters to sediments of microplastic pollution in the southern Mediterranean Sea: A case study of BouIsmail Bay, Algeria. Marine Pollution Bulletin, 222(Pt 2), 118809, https://doi.org/10.1016/j.marpolbul.2025.118809
- Andrady, A.L., 2011. Microplastics in the marine environment. Marine Pollution Bulletin, 62(8), 1596-1605.
- Auta, H., Emenike, C. & Fauziah, S., 2017. Distribution and importance of microplastics in the marine environment: A review of the sources, fate, effects, and potential solutions. Environment International, 102, 165-176, https://doi.org/10.1016/j.envint.2017.02.013
- Boggs, S., 2006. Principles of sedimentology and stratigraphy. New Jersey, USA: Pearson.
- Bouadil, O., Benomar, M., Ouarghi, H. E., Aboulhassan, M.A. & Benbrahim, S. 2024. Identification and quantification of microplastics in surface water of a southwestern Mediterranean Bay (Al Hoceima, Morocco). Waste Management Bulletin, 2(1), 142–151, https://doi.org/10.1016/ j.wmb.2024.01.003
- Boucher, J. & Friot, D., 2017. Primary microplastics in the oceans: A global evaluation of sources. Gland, Switzerland, IUCN, 43pp, https://doi.org/10.2305/ iucn.ch.2017.01.en
- Browne, M.A., Crump, P., Niven, S.J., Teuten, E., Tonkin, A., Galloway, T. & Thompson, R., 2011. Accumulation of microplastic on shorelines worldwide: sources and sinks. Environmental Science & Technology, 45(21), 9175–9179, https://doi.org/10.1021/es201811s
- Camargo, A.L.G., Girard, P., Sanz-Lazaro, C., Silva, A.C.M., De Faria, É., Figueiredo, B.R.S., Caixeta, D.S. & Blettler, M.C.M., 2022. Microplastics in sediments of the Pantanal Wetlands, Brazil. Frontiers in Environmental Science, 10, https://doi.org/ 10.3389/fenvs.2022.1017480
- Claessens, M., De Meester, S., Van Landuyt, L., De Clerck, K. & Janssen, C.R., 2011. Occurrence and distribution of microplastics in marine sediments along the Belgian coast. Marine Pollution Bulletin, 62(10), 2199–2204, https://doi.org/10.1016/j.marpolbul.2011.06.030
- Corcoran, P.L., Biesinger, M.C. & Grifi, M., 2009. Plastics and beaches: A degrading relationship. Marine Pollution Bulletin, 58(1), 80–84, https://doi.org/10.1016/j.marpolbul.2008.08.022
- Cózar, A., Echevarría, F., González-Gordillo, J.I., Irigoien, X., Úbeda, B., Hernández-León, S., Palma, Á.T., Navarro, S., García-De-Lomas, J., Ruiz, A., Fernández-De-Puelles, M.L. & Duarte, C.M., 2014. Plastic debris in the open ocean. Proceedings of the National Academy of Sciences, 111(28), 10239–10244, https://doi.org/10.1073/ pnas.1314705111
- Crawford, C.B. & Quinn, B., 2016. Microplastic identification techniques. In Elsevier eBooks (pp. 219–267), https://doi.org/10.1016/b978-0-12-809406-8.00010-4
- Derraik, J.G., 2002. The pollution of the marine environment by plastic debris: a review. Marine Pollution Bulletin, 44(9), 842–852, https://doi.org/10.1016/s0025-326x(02)00220-5
- Devi, S.S., Gouri, B.R., Anjali, S. & Kumar, A.B., 2024. Microplastic contamination in Ashtamudi Lake, India: Insights from a Ramsar wetland. Journal of Contaminant Hydrology, 264, 104367, https://doi.org/10.1016/j.jconhyd.2024.104367
- Dilshad, A., Taneez, M., Younas, F., Jabeen, A., Rafiq, M.T. & Fatimah, H., 2022. Microplastic pollution in the surface water and sediments from Kallar Kahar wetland, Pakistan: occurrence, distribution, and characterization by ATR-FTIR. Environmental Monitoring and Assessment, 194(7), https://doi.org/10.1007/s10661-022-10171-z
- Eerkes-Medrano, D., Thompson, R.C. & Aldridge, D.C., 2015. Microplastics in freshwater systems: A review of the emerging threats, identification of knowledge gaps and prioritisation of research needs. Water Research, 75, 63–82, https://doi.org/10.1016/j.watres.2015.02.012
- Ellen MacArthur Foundation, 2017. The New Plastics Economy: Rethinking the Future of Plastics & Catalysing Action. World Economic Forum & McKinsey & Company.
- Flores-Cortés, M. & Armstrong-Altrin, J.S., 2022. Textural characteristics and abundance of microplastics in Tecolutla beach sediments, Gulf of Mexico. Environmental Monitoring and Assessment, 194(10).
- Fok, L. & Cheung, P., 2015. Hong Kong at the Pearl River Estuary: A hotspot of microplastic pollution. Marine Pollution Bulletin, 99(1–2), 112–118, https://doi.org/10.1016/j.marpolbul.2015.07.050
- Folk, R.L. & Ward, W.C., 1957. Brazos River bar: A study in the significance of grain size parameters. Journal of Sedimentary Petrology, 27, 3–26.
- Fustec, É. & Lefeuvre, J.-C., 2000. Fonctions et valeurs des zones humides. Paris, France: Dunod, 426 p.
- Gall, S. & Thompson, R., 2015. The impact of debris on marine life. Marine Pollution Bulletin, 92(1–2), 170-179, https://doi.org/10.1016/j.marpolbul.2014.12.041
- Gardel, A., Anthony, E.J., Santos, V.F., Huybrechts, N., Lesourd, S., Sottolichio, A. & Maury, T., 2022. A remote sensing-based classification approach for river mouths of the Amazon-influenced Guianas coast. Regional Environmental Change, 22(2), https://doi.org/10.1007/s10113-022-01913-3
- Gasperi, J., Wright, S.L., Dris, R., Collard, F., Mandin, C., Guerrouache, M., Langlois, V., Kelly, F.J. & Tassin, B., 2017. Microplastics in air: Are we breathing it in? Current Opinion in Environmental Science & Health, 1, 1–5, https://doi.org/10.1016/ j.coesh.2017.10.002
- Gazal, A.A. & Gheewala, S.H., 2020. Plastics, microplastics and other polymer materials – A threat to the environment. Journal of Sustainable Energy & Environment, 11, 113–122.
- Geyer, R., Jambeck, J.R. & Law, K.L., 2017. Production, use, and fate of all plastics ever made. Science Advances, 3(7), https://doi.org/10.1126/ sciadv.1700782
- Grini, H., Metallaoui, S., González-Fernández, D. & Bensouilah, M. 2022. First evidence of plastic pollution in beach sediments of the Skikda coast (northeast of Algeria). Marine Pollution Bulletin, 181, 113831, https://doi.org/10.1016/j.marpolbul.2022.113831
- Gündoğdu, S. & Çevik, C., 2017. Micro- and mesoplastics in Northeast Levantine coast of Turkey: The preliminary results from surface samples. Marine Pollution Bulletin, 118(1–2), 341–347, https://doi.org/10.1016/j.marpolbul.2017.03.002
- Guo, X., Yin, Y., Yang, C. & Dang, Z., 2018. Maize straw decorated with sulfide for tylosin removal from the water. Ecotoxicology and Environmental Safety, 152, 16–23, https://doi.org/10.1016/j.ecoenv.2018.01.025
- Harms, I.K., Diekötter, T., Troegel, S. & Lenz, M., 2021. Amount, distribution and composition of large microplastics in typical agricultural soils in Northern Germany. Science of the Total Environment, 758, 143615.
- Hartmann, N.B., Hüffer, T., Thompson, R.C., Hassellöv, M., Verschoor, A., Daugaard, A.E., Rist, S., Karlsson, T., Brennholt, N., Cole, M., Herrling, M.P., Hess, M.C., Ivleva, N.P., Lusher, A.L. & Wagner, M., 2019. Are we speaking the same language? Recommendations for a definition and Categorization Framework for Plastic Debris. Environmental Science & Technology, 53(3), 1039–1047, https://doi.org/10.1021/acs.est.8b05297
- Hidalgo‑Ruz, V., Gutow, L., Thompson, R.C. & Thiel, M., 2012. Microplastics in the marine environment: A review of the methods used for identification and quantification. Environmental Science & Technology, 46, 3060–3075, https://doi.org/ 10.1021/es2031505
- Ibrahim, Y.S., Hamzah, S.R., Khalik, W.M.A.W.M., Yusof, K.M.K.K. & Anuar, S.T., 2021. Spatiotemporal microplastic occurrence study of Setiu Wetland, South China Sea. The Science of the Total Environment, 788, 147809, https://doi.org/ 10.1016/j.scitotenv.2021.147809
- Jacobs, P. & Ochando, B., 1972. Répartition géographique et importance numérique des anatidés hivernants en Algérie. Le Gerfaut, 69, 239–251.
- Jahan, S., Strezov, V., Weldekidan, H., Kumar, R., Kan, T., Sarkodie, S.A., He, J., Dastjerdi, B. & Wilson, S.P., 2019. Interrelationship of microplastic pollution in sediments and oysters in a seaport environment of the eastern coast of Australia. The Science of the Total Environment, 695, 133924, https://doi.org/10.1016/j.scitotenv.2019.133924
- Jiang, C., Yin, L., Wen, X., Du, C., Wu, L., Long, Y., Liu, Y., Ma, Y., Yin, Q., Zhou, Z. & Pan, H., 2018. Microplastics in sediment and surface water of West Dongting Lake and South Dongting Lake: Abundance, source and composition. International Journal of Environmental Research and Public Health, 15(10), 2164, https://doi.org/10.3390/ ijerph15102164
- Jian-Wu, L., Wei, Y., Gan-Lin, Z., Li-Dong, Z., Yong-Jian, J. & Zi-Tong, G., 2013. Grain size evidence of multiple origins of red clays in the Jinhua-Quzhou Basin, South China. Pedosphere, 23, 686–695.
- Jiwarungrueangkul, T., Phaksopa, J., Sompongchaiyakul, P. & Tipmanee, D., 2021. Seasonal microplastic variations in estuarine sediments from urban canal on the west coast of Thailand: A case study in Phuket province. Marine Pollution Bulletin, 168, 112452, https://doi.org/ 10.1016/j.marpolbul.2021.112452
- Karlsson, T.M., Arneborg, L., Broström, G., Almroth, B.C., Gipperth, L. & Hassellöv, M., 2018. The unaccountability case of plastic pellet pollution. Marine Pollution Bulletin, 129(1), 52–60, https://doi.org/10.1016/j.marpolbul.2018.01.041
- Khazr, A., Ali, M., Samir, G., Amel, H., Sondes, M., Asma, H.S., Mustapha, B., Samir, T., Fatma, G., Mourad, B., Badreddine, S., Mohamed, B., Mohamed, D., Ezzeddine, M. & Beyrem, H., 2025. Assessment of microplastic pollution: distribution, composition, and sources in the El-Mellah (Algeria) and Bizerte (Tunisia) Mediterranean lagoons. Journal of Environmental Science and Health Part A, 60(9), 495–506, https://doi.org/10.1080/10934529.2025.2605863
- Klein, S., Worch, E. & Knepper, T.P., 2015. Occurrence and spatial distribution of microplastics in river shore sediments of the Rhine-Main area in Germany. Environmental Science & Technology, 49(10), 6070–6076, https://doi.org/10.1021/acs.est.5b00492
- Lassen, C., Foss Hansen, S., Magnusson, K., Norén, F., Bloch Hartmann, N.I., Rehne Jensen, P., Gissel Nielsen, T. & Brinch, A., 2015. Microplastics—occurrence, effects and sources of releases to the environment in Denmark. Environmental Project No. 1793. Copenhagen: Environment Protection Agency, Ministry of Environment and Food of Denmark.
- Law, K.L., Morét-Ferguson, S., Maximenko, N.A., Proskurowski, G., Peacock, E.E., Hafner, J. & Reddy, C.M., 2010. Plastic accumulation in the North Atlantic subtropical gyre. Science, 329(5996), 1185–1188, https://doi.org/10.1126/ science.1192321
- Ledant, J.-P. & Vandijk, G., 1977. Situation des zones humides algériennes et de leur avifaune. Aves, 14, 217–232.
- Li, J., Zhang, H., Zhang, K., Yang, R., Li, R. & Li, Y., 2018. Characterization, source, and retention of microplastic in sandy beaches and mangrove wetlands of the Qinzhou Bay, China. Marine Pollution Bulletin, 136, 401–406, https://doi.org/ 10.1016/j.marpolbul.2018.09.025
- Li, L., Li, M., Deng, H., Cai, L., Cai, H., Yan, B., Hu, J. & Shi, H., 2018. A straightforward method for measuring the range of apparent density of microplastics. The Science of the Total Environment, 639, 367–373, https://doi.org/ 10.1016/j.scitotenv.2018.05.166
- Li, N., Wu, M., Zhang, Y., Yuan, W., Wu, J. & Shao, X., 2022. A review on microplastics pollution in coastal wetlands. Watershed Ecology and the Environment, 5, 24–37, https://doi.org/10.1016/ j.wsee.2022.11.010
- Lots, F.A., Behrens, P., Vijver, M.G., Horton, A.A. & Bosker, T., 2017. A large-scale investigation of microplastic contamination: Abundance and characteristics of microplastics in European beach sediment. Marine Pollution Bulletin, 123(1–2), 219–226, https://doi.org/10.1016/j.marpolbul.2017.08.057
- Luo, W., Su, L., Craig, N.J., Du, F., Wu, C. & Shi, H., 2019. Comparison of microplastic pollution in different water bodies from urban creeks to coastal waters. Environmental Pollution, 246, 174–182.
- Lusher, A.L., Bråte, I.L.N., Munno, K., Hurley, R.R. & Welden, N.A., 2020. Is it or isn't it: the importance of visual classification in microplastic characterization. Applied Spectroscopy, 74(9), 1139–1153.
- Lusher, A.L., Burke, A., O’Connor, I. & Officer, R., 2014. Microplastic pollution in the Northeast Atlantic Ocean: Validated and opportunistic sampling. Marine Pollution Bulletin, 88(1–2), 325–333, https://doi.org/10.1016/j.marpolbul.2014.08.023
- Maltby, E. & Barker, T., 2009. The Wetlands Handbook. Wiley-Blackwell. Edited by Edward Maltby and Tom Barker. ISBN: 978-0-632-05255-4
- Mani, H., Khunti, K., Daly, H., Barnett, J. & Davies, M., 2015. Education and self-management for women with polycystic ovary syndrome; a narrative review of literature. Ibnosina Journal of Medicine and Biomedical Sciences, 07(01), 1–9, https://doi.org/10.4103/1947-489x.210263
- Mendes, A.M., Golden, N., Bermejo, R. & Morrison, L., 2021. Distribution and abundance of microplastics in coastal sediments depends on grain size and distance from sources. Marine Pollution Bulletin, 172, 112802.
- Menges, G., Michaeli, W. & Mohren, P., 2001. How to make injection molds (3rd ed.). Hanser Publishers.
- Mercy, F.T., Alam, A.R. & Akbor, M.A., 2022. Abundance and characteristics of microplastics in major urban wetlands of Dhaka, Bangladesh. Research Square, https://doi.org/10.21203/rs.3.rs-1455552/v1
- Metzmacher, M., 1979. Les oiseaux de la Macta et de sa région (Algérie): Non passereaux. Aves, 3–4, 89–123.
- Mitsch, W.J. & Gosselink, J.G., 2007. Wetlands (4th Edition). John Wiley & Sons, Inc., Hoboken.
- Naidoo, T., Glassom, D. & Smit, A.J., 2015. Plastic pollution in five urban estuaries of KwaZulu-Natal, South Africa. Marine Pollution Bulletin, 101(1), 473–480, https://doi.org/10.1016/j.marpolbul.2015.09.044
- NAW, 2020. National Agency of Waste, the State of Waste Management in Algeria, p. 150.
- NAW, 2021. National Agency of Waste, Plastic waste in Algeria. Cross-examination of single-use plastics, pp. 26.
- Nor, N.H.M. & Obbard, J.P., 2014. Microplastics in Singapore’s coastal mangrove ecosystems. Marine Pollution Bulletin, 79(1–2), 278–283, https://doi.org/10.1016/j.marpolbul.2013.11.025
- Pérez-Alvarado, B.Y. & Armstrong-Altrin, J.S., 2025. Microplastics in the Barra Norte and Mocambo beach sediments, Gulf of Mexico, Mexico. Carpathian Journal of Earth and Environmental Sciences, 20(2), 241–254, https://doi.org/10.26471/ cjees/2025/020/329
- Plastics Europe, 2020. Plastics - the Facts 2020: An analysis of European plastics production, demand and waste data.
- Pradit, S., Noppradit, P., Loh, P., Nitiratsuwan, T., Le, T.P.Q., Oeurng, C., Mohamed, C.A.R., Lee, C.W., Lu, X., Anshari, G.Z., Kandasamy, S. & Wang, J., 2022. The Occurrence of Microplastics in Sediment Cores from Two Mangrove Areas in Southern Thailand. Journal of Marine Science and Engineering, 10(3), 418, https://doi.org/10.3390/jmse10030418
- Prata, J.C., Silva, A.L., Walker, T.R., Duarte, A.C. & Rocha-Santos, T., 2020. COVID-19 pandemic repercussions on the use and management of plastics. Environmental Science & Technology, 54(13), 7760–7765, https://doi.org/10.1021/acs.est.0c02178
- Qian, J., Tang, S., Wang, P., Lu, B., Li, K., Jin, W. & He, X., 2020. From Source to Sink: Review and Prospects of Microplastics in Wetland ecosystems. The Science of the Total Environment, 758, 143633, https://doi.org/10.1016/j.scitotenv.2020.143633
- Rahmani, R., Ghannem, S., Zahouani, Y., Khazri, A., Kanzari, S., Bilal, E. & Touaylia, S., 2025. Microplastics in a lotic freshwater environment: Typology and profile of occurrence along Joumine Stream, affluent of the Ichkeul Wetland (Northern Tunisia). Carpathian Journal of Earth and Environmental Sciences, 20(1), 85–95, https://doi.org/10.26471/cjees/2025/020/316
- Ramos-Vázquez, M.A., Verma, S.K., Armstrong-Altrin, J.S., James, R.A. & Madhavaraju, J., 2024. Characterization of microplastics in marine sediments from the Gulf of Tehuantepec, Mexican Pacific. Carpathian Journal of Earth and Environmental Sciences, 19(2), 255–264, https://doi.org/10.26471/cjees/2024/019/296
- Rasta, M., Sattari, M., Taleshi, M.S. & Namin, J.I., 2020. Identification and distribution of microplastics in the sediments and surface waters of Anzali Wetland in the Southwest Caspian Sea, Northern Iran. Marine Pollution Bulletin, 160, 111541, https://doi.org/10.1016/j.marpolbul.2020.111541
- Reynolds, C. & Ryan, P.G., 2018. Micro-plastic ingestion by waterbirds from contaminated wetlands in South Africa. Marine Pollution Bulletin, 126, 330–333.
- Rillig, M.C., Ziersch, L. & Hempel, S., 2017. Microplastic transport in soil by earthworms. Scientific Reports, 7(1), https://doi.org/10.1038/s41598-017-01594-7
- Rochman, C.M., Browne, M.A., Halpern, B.S., Hentschel, B.T., Hoh, E., Karapanagioti, H.K., Rios-Mendoza, L.M., Takada, H., Teh, S. & Thompson, R.C., 2013. Classify plastic waste as hazardous. Nature, 494(7436), 169–171, https://doi.org/10.1038/494169a
- Rodrigues, C., Rodrígues, Y., Frias, J., Carriço, R., Sobral, P., Antunes, J., Duncan, E.M. & Pham, C.K., 2024. Microplastics in beach sediments of the Azores archipelago, NE Atlantic. Marine Pollution Bulletin, 201, 116243.
- Rosato, D.V. & Rosato, M.G., 2001. Plastics Design Handbook. Springer.
- Russell, S.J., 2007. Handbook of nonwovens. Woodhead Publishing Limited eBooks, https://doi.org/ 10.1533/9781845691998
- Ryan, P.G., 2013. Litter survey detects the South Atlantic ‘garbage patch’. Marine Pollution Bulletin, 79(1–2), 220–224, https://doi.org/10.1016/j.marpolbul.2013.12.010
- Setälä, O., Fleming-Lehtinen, V. & Lehtiniemi, M., 2014. Ingestion and transfer of microplastics in the planktonic food web. Environmental Pollution, 185, 77–83.
- Setiti, S., Hamdi, B., Chernai, S., Bachari, F.H., Bachouche, S., Ghezali, Y. & Suaria, G., 2021. Seasonal variation of microplastics density in Algerian surface waters (South-Western Mediterranean Sea). Mediterranean Marine Science, https://doi.org/10.12681/mms.24899
- Strong, A.B., 2006. Plastics: Materials and processing (3rd ed.). Pearson Prentice Hall.
- Su, L., Xue, Y., Li, L., Yang, D., Kolandhasamy, P., Li, D. & Shi, H., 2016. Microplastics in Taihu Lake, China. Environmental Pollution, 216, 711–719, https://doi.org/10.1016/j.envpol.2016.06.036
- Szcześniak, M., Kokociński, M., Jagodziński, R., Pleskot, K., Zajączkowski, M. & Szczuciński, W., 2023. Late Holocene Vistula River floods recorded in grain size distributions and diatom assemblages of marine sediments of the Gulf of Gdańsk (Baltic Sea). Palaeogeography, Palaeoclimatology, Palaeoecology, 617, 111499, https://doi.org/10.1016/j.palaeo.2023.111499
- Taïbi, N., Bentaallah, M. E. A., Alomar, C., Compa, M., & Deudero, S. 2021. Micro- and macro-plastics in beach sediment of the Algerian western coast: First data on distribution, characterization, and source. Marine Pollution Bulletin, 165, 112168, https://doi.org/10.1016/j.marpolbul.2021.112168
- Tanabe, S., Komatsubara, T. & Hori, K., 2023. Grain-size variability and formation process of lowstand river sediments in the Japanese Islands: A review and outlook. Earth-Science Reviews, 243, 104504, https://doi.org/10.1016/j.earscirev.2023.104504
- Thompson, R.C., Olsen, Y., Mitchell, R.P., Davis, A., Rowland, S.J., John, A.W.G., McGonigle, D. & Russell, A.E., 2004. Lost at sea: Where is all the plastic? Science, 304(5672), 838, https://doi.org/ 10.1126/science.1094559
- Tnoumi, A., El Khalidi, K., Zourarah, B., Angelone, M., Armiento, G., Caprioli, R., Crovato, C., De Cassan, M., Montereali, M.R., Parrella, L., Proposito, M., Spaziani, F. & Nardi, E., 2020. Studies on the textural characteristics of sediments from Khnifiss Lagoon (southern coast of Morocco). International Journal of Advanced Research in Engineering and Technology, 11(12), 2299–2311, https://doi.org/10.34218/IJARET.11.12.2020.218
- Turner, R.K., 1992. Les défaillances du marché et des gouvernements dans la gestion de l'environnement: les zones humides et les forêts. OCDE.
- Uddin, S., Fowler, S.W., Uddin, M.F., Behbehani, M. & Naji, A., 2021. A review of microplastic distribution in sediment profiles. Marine Pollution Bulletin, 163, 111973, https://doi.org/10.1016/ j.marpolbul.2021.111973
- Véla, E. & Benhouhou, S., 2007. Évaluation d’un nouveau point chaud de biodiversité végétale dans le Bassin méditerranéen (Afrique du Nord). Comptes Rendus Biologies, 330(8), 589–605, https://doi.org/10.1016/j.crvi.2007.04.006
- Vianello, A., Boldrin, A., Guerriero, P., Moschino, V., Rella, R., Sturaro, A. & Da Ros, L., 2013. Microplastic particles in sediments of Lagoon of Venice, Italy: First observations on occurrence, spatial patterns and identification. Estuarine Coastal and Shelf Science, 130, 54–61, https://doi.org/10.1016/j.ecss.2013.03.022
- Wang, C., Zhao, J. & Xing, B., 2020. Environmental source, fate, and toxicity of microplastics. Journal of Hazardous Materials, 407, 124357, https://doi.org/10.1016/j.jhazmat.2020.124357
- Wang, J., Peng, J., Tan, Z., Gao, Y., Zhan, Z., Chen, Q. & Cai, L., 2017. Microplastics in the surface sediments from the Beijiang River littoral zone: Composition, abundance, surface textures and interaction with heavy metals. Chemosphere, 171, 248–258, https://doi.org/10.1016/j.chemosphere.2016.12.074
- Williams, M. (Ed.), 1990. Wetlands: A threatened landscape. Oxford, UK: Blackwell.
- Woodall, L.C., Gwinnett, C., Packer, M., Thompson, R.C., Robinson, L.F. & Paterson, G.L., 2015. Using a forensic science approach to minimize environmental contamination and to identify microfibres in marine sediments. Marine Pollution Bulletin, 95(1), 40–46, https://doi.org/10.1016/j.marpolbul.2015.04.044
- Xu, S., Ma, J., Ji, R., Pan, K. & Miao, A., 2019. Microplastics in aquatic environments: Occurrence, accumulation, and biological effects. The Science of the Total Environment, 703, 134699, https://doi.org/10.1016/j.scitotenv.2019.134699
- Yang, T., Gao, M. & Nowack, B., 2022. Formation of microplastic fibers and fibrils during abrasion of a representative set of 12 polyester textiles. The Science of the Total Environment, 862, 160758, https://doi.org/10.1016/j.scitotenv.2022.160758
- Yin, L., Wen, X., Du, C., Jiang, J., Wu, L., Zhang, Y., Hu, Z., Hu, S., Feng, Z., Zhou, Z., Long, Y. & Gu, Q., 2020. Comparison of the abundance of microplastics between rural and urban areas: A case study from East Dongting Lake. Chemosphere, 244, 125486, https://doi.org/10.1016/j.chemosphere.2019.125486
- Zhang, D., Fraser, M.A., Huang, W., Ge, C., Wang, Y., Zhang, C. & Guo, P., 2020. Microplastic pollution in water, sediment, and specific tissues of crayfish (Procambarus clarkii) within two different breeding modes in Jianli, Hubei province, China. Environmental Pollution, 272, 115939, https://doi.org/10.1016/j.envpol.2020.115939
- Zhang, K., Gong, W., Lv, J., Xiong, X. & Wu, C., 2015. Accumulation of floating microplastics behind the Three Gorges Dam. Environmental Pollution, 204, 117–123, https://doi.org/10.1016/j.envpol.2015.04.023
- Zhang, K., Su, J., Xiong, X., Wu, X. & Wu, C., 2020. Microplastic pollution in surface water and sediments of two typical estuaries in China. Environmental Pollution, 244, 965–973.
- Zobkov, M., Belkina, N., Kovalevski, V., Zobkova, M., Efremova, T. & Galakhina, N., 2020. Microplastic abundance and accumulation behavior in Lake Onego sediments: a journey from the river mouth to pelagic waters of the large boreal lake. Journal of Environmental Chemical Engineering, 8(5), 104367, https://doi.org/ 10.1016/j.jece.2020.104367
